
日前,艾德生物自主研发的KRAS/NRAS基因突变联合检测试剂盒和KRAS/NRAS/BRAF基因突变联合检测试剂盒已获得国家药监局(CFDA)批准上市,填补了国内市场空白。
至此,针对晚期以及转移或复发结直肠癌患者的抗EGFR靶向治疗前的用药检测,艾德已形成完善的产品线:
1、KRAS基因突变检测试剂盒(可检测KRAS外显子2、3、4上的多种KRAS基因突变类型)---CFDA批准(国械注准20153401126)、CE认证
2、NRAS基因突变检测试剂盒(可检测NRAS外显子2、3、4上的多种NRAS基因突变类型)---CFDA批准(国械注准20153401125)、CE认证
3、KRAS/NRAS基因突变联合检测试剂盒(可同时检测KRAS和NRAS外显子2、3、4的多种基因突变类型)---CFDA批准(国械注准20153401885)、CE认证
4、KRAS/NRAS/BARF基因突变联合检测试剂盒(可同时检测KRAS和NRAS外显子的2、3、4以及BRAF外显子15的多种基因突变类型)---CFDA批准(国械注准20153401886)、CE认证
5、KRAS/NRAS/BARF/PIK3CA基因突变联合检测试剂盒(可同时检测KRAS和NRAS的外显子2、3、4、BRAF 外显子15以及PIK3CA外显子20的多种基因突变类型)---CFDA批准(国械注准20153401124)
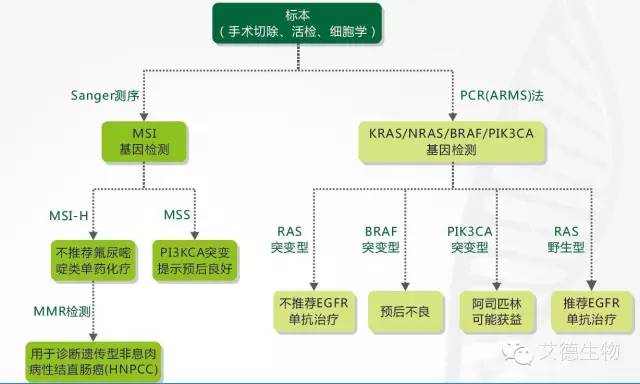
AmoyDx为结直肠癌精准诊治提供系列解决方案
艾德生物结直肠癌癌靶向检测系列产品的特点:
1、首批获得CFDA批准上市的产品
2、最齐全的结直肠癌靶向基因检测产品线
3、采用临床认可的ADx-ARMS技术
4、检测所覆盖的位点符合临床需求
5、只需1-3片结直肠癌肿瘤组织切片或活检组织
6、可在4.5小时内完成对肿瘤组织标本的提取和检测
7、适用于主流的各型PCR仪。
结直肠癌临床指南和专家共识摘录(基因检测)
一、NCCN结直肠癌临床实践指南(2015 V2)[1,2]
1、所有转移性结直肠癌患者都应进行RAS基因检测(KRAS和NRAS)。只要有KRAS基因突变(外显子2或者其他外显子)或NRAS基因突变,西妥昔单抗及帕尼单抗就不再适用于此类患者的治疗。
2、具有V600E BRAF突变的患者,似乎预后更差。尚缺乏足够的数据,依据患者的V600E BRAF突变状态来指导抗EGFR单抗与一线治疗中有效化疗方案的联合使用。现时有限的资料提示,患者存在V600E突变时,一线治疗进展后使用抗EGFR单抗治疗是无效的。
3、检测可采用福尔马林固定、石蜡包埋的组织。所取组织可以是原发结直肠癌组织和/或转移灶,有文献报道两种标本的KRAS突变、NRAS突变和BRAF突变情况相似。
4、具有MSI-H的Ⅱ期患者可能预后更好,不会从5-FU单药辅助化疗中获益。
二、2015ASCO结直肠癌分子检测指南[3]
建议对结直肠癌的分子标记检测应包括以下内容:
1、应该对考虑行抗EGFR治疗的患者,进行结直肠癌组织的RAS突变检测。这种检测必须包括KRAS和NRAS外显子2的密码子12、13;外显子3的密码子59、61,和外显子4的密码子117、146。
2、BRAF V600突变检测,应与缺陷错配修复(dMMR)/微卫星不稳定性(MSI)同时进行,用来进行预后分层。
关于结直肠癌分子标记检测最合适的样品:
1、用原发结直肠癌组织来进行分子标记物检测(KRAS,扩展RAS,BRAF和dMMR / MSI)是可以接受的。如果患者出现转移,用转移组织来行相关的分子检测,这也是可以接受的。
2、福尔马林固定的石蜡包埋组织是一个可接受的样本。使用其他标本将需要额外的足够的证据。组织处理过程的任何改变都需要经过相似的验证。
三、结直肠癌诊疗规范(2015年版)[4]
1、确定为复发或转移性结直肠癌时,推荐检测KRAS、NRAS和BRAF 基因状态。
2、对于Ⅱ期结肠癌患者建议进行MMR蛋白表达或MSI检测,存在MMR蛋白表达缺失或MSI-H的患者,不推荐氟尿嘧啶单药辅助化疗。
Tabernero 博士[5]的一项多中心 I 期研究表明,BRAF抑制剂+抗EGFR治疗+PIK3CA抑制剂的结直肠癌联合治疗生存获益与标准治疗相比较可以使患者的无病生存延长2倍。
参考文献:
1.NCCN Guidelines Version2.2015- Colon cancer
2.NCCN Guidelines Version2.2015- Rectalcancer
3.Roxanne Nelson, New Guidelines on Colorectal Cancer Molecular Testing,April 02, 2015
4.中国结直肠癌诊疗规范(2015版)
5. Cancer Discovery February 2015 5;102.doi:10.1158/2159-8290.CD-NB2014-186
关于艾德
厦门艾德生物医药科技股份有限公司是专业化的肿瘤个体化分子诊断产品研发及生产企业,致力于肿瘤个体化分子诊断技术的创新和高端分子诊断产品的产业化,是细分市场的领先企业。
公司研发的ADx-ARMS®技术拥有完全自主知识产权,是目前国际上个体化分子诊断领域最先进的技术之一,达到行业公认的国际领先水平。基于核心技术,艾德生物已研发出20余种肿瘤个体化分子诊断产品,其中EGFR、KRAS、NRAS、KRAS/NRAS、BRAF、KRAS/NRAS/BRAF、EML4-ALK、PIK3CA、ALK/ROS1、ROS1、KRAS/NRAS/BRAF/PIK3CA、HER-2十二种目前在肿瘤个体化分子诊断领域最重要的基因突变检测产品率先获得CFDA《医疗器械注册证》和欧盟CE认证。目前,艾德产品已在市场大规模销售,涵盖国内大多数三甲医院和40多个国家和地区。
未来,艾德生物将和中国的临床专家和病理专家更紧密地合作,为中国的分子诊断提供更好的服务。