OriginCell Therapeutics Presents Positive Results of GPC3 CAR-T in HCC Trial at the 2021 ASCO Annual Meeting: Company
Brief: Qiming’s portfolio company OriginCell Therapeutics presented the updated data from the on-going trial of Ori-CAR-001, a GPC3 CAR-T cell therapy for the treatment of relapsed/refractory hepatocellular carcinoma (HCC), at the 2021 annual meeting of American Society of Clinical Oncology (ASCO). Preliminary results show promising safety and efficacy. Preliminary results on the efficacy and safety were promising.
Below is the company’s press release.
SHANGHAI, June 6, 2021 /PRNewswire/ -- On June 4, 2021 (EST), OriginCell Therapeutics (Shanghai) Co., Ltd. ("OriginCell"), together with Lishui Central Hospital affiliated to Zhejiang University and Shanghai Changzheng Hospital, presented the updated data from the on-going trial of Ori-CAR-001, a GPC3 CAR-T cell therapy for the treatment of relapsed/refractory hepatocellular carcinoma (HCC), at the 2021 annual meeting of American Society of Clinical Oncology. Preliminary results from the study show promising safety and efficacy of Ori-CAR-001 in patients with GPC3-positive relapsed/refractory HCC.
Session type: Poster Session
Abstract Title: An armored GPC3-directed CAR-T for refractory or relapsed hepatocellular carcinoma in China: A phase I trial
Date and time: 06/04/2021, 9:00 AM (EDT)
Abstract ID: 4095
Link: https://meetinglibrary.asco.org/record/199036/abstract
A breakthrough Cell Therapy against Solid Tumors
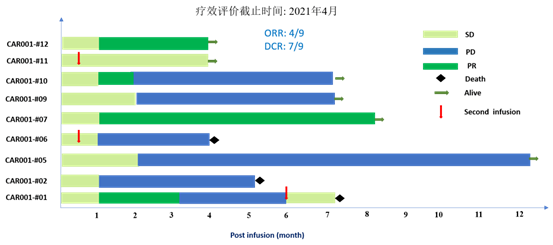
As of March 10, 2021, a total of 11 patients were enrolled into this study. All subjects had advanced HCC and had failed prior lines of treatment including chemotherapy, TACE, and targeted therapies. All subjects had BCLC stage C (late stage) HCC except for one subject with stage B (mid stage). All subjects had multiple lesions, of which 6 (54%) had distant metastasis. Two subjects dropped out early and were not evaluable. Among the 9 evaluable subjects, 4 achieved partial response (PR), 3 achieved stable disease (SD), and 2 had disease progression (PD). The objective response rate was 44.4%, and the disease control rate reached 77.8%. A partial response lasting for more than 6 months was observed in Subject 007 at the time of data cutoff, whose tumor volume was reduced by more than 80% one month post cell therapy infusion. (As of June 2021, this subject had exceeded 8 months of disease control, follow-up is still ongoing). Good tumor response was also seen in Subject 012, who has advanced and diffuse massive HCC and joined the study after failing in radiotherapy, targeted drug and 12 times of TACE.
Barcelona Clinic Liver Cancer (BCLC) is a liver cancer staging system developed by the European Association for the Study of Liver, to help assess patients' conditions, select the right therapies for the right patients, and predict patients' prognosis. The BCLC system classifies HCC mainly as stages of 0, A, A1,A2,A3,A4, B, C,and D.
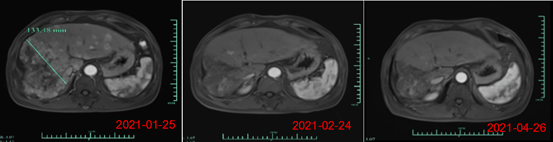
On day 28 post infusion with CAR-T cells, the target tumor lesion decreased from 133mm in diameter at baseline to 9mm, a reduction of more than 93%. Currently, the subject is being evaluated for month 3 post CAR-T cell infusion, MRI scan showed near complete tumor response. In addition, AFP level (alpha-fetoprotein, a specific tumor marker for primary liver cancer) decreased from a baseline value of >80,000/ng/ml to 1148.9 ng/ml on month 1, and to 746.7 ng/ml at month 3 post infusion (normal range <40ng/ml), demonstrating significant preliminary efficacy of Ori-CAR-001 in patients with advanced HCC.
Good Safety Profile
Ori-CAR-001 CAR-T infusion demonstrated good safety and tolerability in 11 patients. Cytokine release syndrome (CRS) was observed in 9 patients (7 cases grade 1-2 and 2 cases grade 4), which were resolved after treatment with steroids and tocilizumab. No neurotoxicity was observed.
Regarding the results from this exploratory study, Professor Ji Jiansong, principal investigator of Lishui Central hospital, who is also Chief scientist of the National Key R&D Program, director of Key Laboratory of Imaging Diagnosis and Minimally Invasive Intervention Research of Zhejiang China, said, "There is currently no effective treatment for relapsed/refractory hepatocellular carcinoma. Many challenges such as the tumor micro environment is difficult to overcome and have limited the therapeutic effects of cell therapy in solid tumors. However, Ori-CAR-001, developed by OriginCell Therapeutics, has managed to achieve an objective response rate of 44% and a disease control rate of 77% in patients with relapsed/refractory HCC, demonstrating a level of safety and anti-tumor activity that cannot be achieved by currently available drugs. Given its preliminary promising efficacy and safety data, we are confident in the future development prospects of Ori-CAR-001 and will continue our research studies. We take great pride and hope Ori-CAR-001 will one day become a new treatment option for patients with advanced HCC.
Dr. He Xiaowen, co-founder and chief scientific officer of OriginCell Therapeutics, said, " Although Immuno-cell therapies have produced remarkable clinical responses in hematological tumors, they have limited efficacy against solid tumors. OrginCell will continue to explore the broader unmet needs in this space, with the hope to bring more effective immuno-cell therapies to clinicians and patients with advanced stage cancer."
About Ori-CAR-001
Ori-CAR-001 is a next generation autologous chimeric antigen receptor T cell therapy targeting GPC3 for the treatment of refractory/ relapsed hepatocellular carcinoma (HCC). The construct of Ori-CAR-001 is comprised of humanized anti-GPC3 scFv, a CD8-derived hinge region, a transmembrane domain linked to 4-1BB co-stimulatory domain, a CD3z intracellular signaling domain, and Ori2 components simultaneously expressed through T2A. Compared with conventional CAR-T cells, Ori-CAR-001 has significantly higher proportion of memory stem T cells, which may translate to improved expansion and persistence in the body, thereby increasing their anti-tumor activity.
It is worth noting that, in addition to the preliminary safety and efficacy data of Ori-CAR-001 presented at 2021 ASCO annual meeting, OriginCell and study investigators also carried out translational medicine studies, in which the mode of administration, dosing, PK/PD, and biomarkers were explored. This will futher promote the application and development of cell therapy for solid tumors.
OriginCell will plan more clinical development programs to further explore Ori-CAR-001's potential based on existing data, and the Investigational New Drug (IND) application with the National Medical Products Administration (NPMA) is currently in progress.
About ASCO
With more than 40,000 members from more than 100 countries, the American Society of Clinical Oncology (ASCO) is the world's leading academic organization for physicians and oncology professionals caring for people with cancer. Its mission is to prevent cancer and improve cancer care, and promote the translation of research findings into clinical practice. By providing access to a global network of oncology expertise, the ASCO annual meeting is recognized as the world's most influential academic congress in the field of oncology.
About OriginCell Therapeutics
Founded in 2015, OriginCell Therapeutics Co., Ltd. ( Chinese name changed from "原能医学"to "原启生物" on May 25) received pre-Series A funding of nearly 100 million yuan exclusively from Qiming Venture Capital at the end of 2019, and the company completed Series A funding of more than 200 million yuan at the end of 2020. With the support of Qiming Venture Capital, OriginCell has been spun off from the OriginCell Group as an innovative drug research and development asset, and will focus on developing new immunotherapies for cancer treatment in the future.
Dedicated to independent innovation, OriginCell has built four innovative and synergistic proprietary technology platforms, i.e. Ori™Ab, an antibody development and optimization technology platform; Ori™CAR, a high-memory CAR T technology platform; Ori ™TIL, an efficient cell expansion and culture technology platform; and Ori™UCAR, a universal CAR-T technology platform.
About Ori™Ab—Antibody Discovery Technology Platform
Ori™Ab is a technology platform that enables the expression of 1011 fully human antibody phage display libraries, 1012 nano antibody libraries, and adds antibody engineering capabilities. A series of superior antibody drug candidates and CAR-T cell target antibodies have been produced from this platform.
About Ori™CAR Technology Platform
Ori™CAR enables the insertion of unique signaling domains into novel and proprietary CAR construct, which greatly enhances the expansion efficiency of memory immune cells, effectively resists the stressful immune environment of solid tumors, enhances the efficacy and persistence of CAR-T, and effectively inhibits tumor growth and prevents recurrence.
About Ori™TIL—A Cell Expansion and Culture Technology Platform
Ori™TIL can substantially increase the number of expanded TIL cells, improve quality attributes, and process parameters to break the therapeutic limitations. The clinical efficacy and safety can be ensured via younger TIL cells that can bring enhanced anti-tumor activity.
About Ori™ UCAR Technology Platform
Ori™ UCAR can efficiently synergize with antibody technology, Ori technology, gene editing, and cell differentiation technology to make future CAR-T therapies more universal and convenient, while effectively reducing its cost of use.
SOURCE OriginCell Therapeutics Co., Ltd.