在2024年美国糖尿病协会(ADA)第84届科学会议上,启明创投投资企业甘李药业(603087.SH)首次公布了GZR18注射液的1b/2a临床研究突破性进展(Late Breaking)结果(摘要编号:1858-LB),即创新型GLP-1受体激动剂GZR18注射液在一项1b/2a期研究中使肥胖受试者减轻了18.6%的体重。
GZR18注射液1b/2a期研究主要研究者、北京大学人民医院纪立农主任担任本次壁报展示讲者
本次研究的关键结果显示:GZR18注射液每周一次(QW)和每两周一次给药(Q2W)治疗35周,在中国肥胖症受试者中平均体重减轻17.8%(QW)与12.8%(Q2W),相较安慰剂组(体重增加0.7%)分别减轻了18.6%与13.5%。此外,GZR18注射液还可改善受试者整体代谢情况。与同类药品相似给药周期的3期临床试验结果相比,本研究的GZR18注射液减重效果超过了司美格鲁肽2.4 mg(9.8%)和替尔泊肽15 mg(17.5%)在相同可比人群的研究数据。这表明,单靶点胰高血糖素样肽-1受体激动剂(GLP-1RA)双周制剂在治疗肥胖症方面的疗效可能与多靶点GLP-1RA类药物相当。该研究结果显示出GZR18注射液具有在每周一次和每两周一次给药频率下提供优于多靶点GLP-1RA类药物减重疗效的潜力。
过去20年,中国超重/肥胖及相关慢性病患病率迅速攀升。研究预测,至2030年,中国成人(≥18岁)超重/肥胖率将达65.3%,儿童青少年(<18岁)将达47.4%。GLP-1RA因其减重效果显著,广泛用于临床体重管理。甘李药业研发的新型GLP-1RA GZR18注射液在临床前研究中已表现出每周一次给药的可能性,并可显著降低动物体重。此次在ADA年会上公布的最新1b/2a期临床研究进一步评估了GZR18注射液在中国肥胖成人中的有效性和安全性。
01/
GZR18注射液的分子设计及延长作用机制
GZR18注射液是一种基于脂肪酸酰化设计的GLP-1RA,具有每周或每两周给药一次的潜力,其与人源GLP-1分子的同源性高达94%。GZR18注射液在人源GLP-1肽链基础上进行了3处氨基酸位点修饰,从而将脂肪酸酰化选择性限制到第26位赖氨酸上。GZR18注射液选用22碳脂肪二酸侧链,与白蛋白强力且可逆结合,从而延长作用时间。多肽主链和脂肪酸侧链由2个OEG(3,8-二氧代氨基辛酸)间隔子和1个L-γ谷氨酸连接子连接,使GZR18注射液的肽链在体内与白蛋白的物理距离更近,利于空间折叠,降低药物降解,延长半衰期。GZR18注射液独特的分子设计优化其与GLP-1受体的亲和力,减少GLP-1受体介导的胃肠道反应,提高药物耐受性。给予适当的滴定给药方案,GZR18注射液有望实现更高的治疗剂量,以期实现更好的治疗效果。
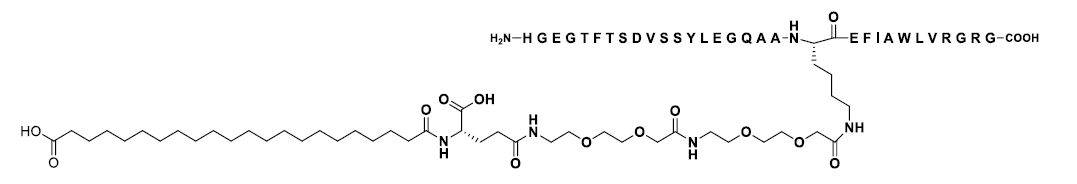
GZR18注射液的分子设计
02/
GZR18注射液中国1b/2a期试验设计
关键纳入标准
试验设计
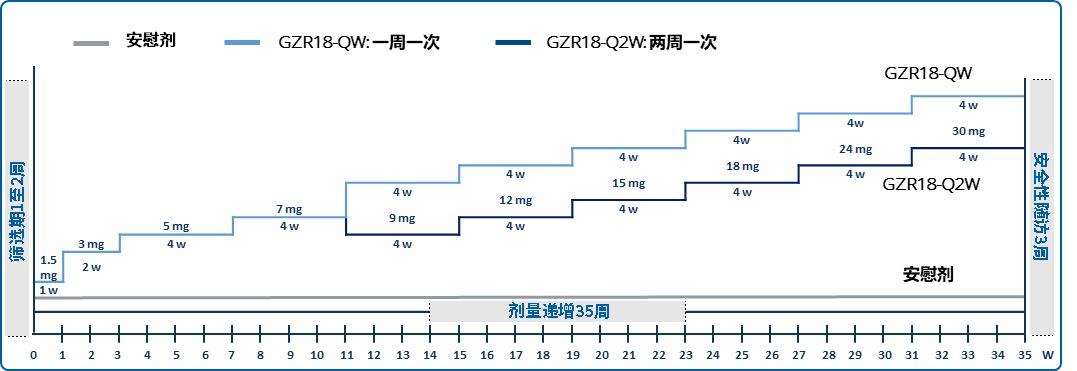
试验信息
研究终点
03/
主要研究结果
GZR18注射液在中国肥胖受试者中的减重疗效优于安慰剂。治疗35周后,GZR18注射液QW组体重较基线的平均变化差值为-16.5 kg(95%CI:-19.9 kg, -13.1 kg);体重与安慰剂组的差值达-18.6%(95%CI:-25.5%, -11.6%)。同时,GZR18注射液Q2W组体重较基线的平均变化差值为-11.3 kg(95%CI:-15.4 kg, -7.2 kg);与安慰剂组的平均变化差值达-13.5%(95%CI:-21.0%, -6.0%)。
GZR18注射液在肥胖受试者中表现出良好的安全性和耐受性。治疗期间最常见的不良事件(AE)为胃肠道反应,多呈轻至中度,与GLP-1RA类药物的常见反应一致,且主要发生在剂量爬坡早期阶段。本研究未发生严重低血糖事件,未发生与试验用药品有关的严重不良事件。
GZR18注射液治疗后BMI与腰围降低幅度优于安慰剂组,且能够改善心脏代谢风险因素。
03/
讨论与未来
GLP-1RA类药物靶点并非越多越好
近年,科学家在不断研究不同减重靶点的组合,例如双重或三重肠促胰素受体激动剂或拮抗剂(如GLP-1R与GIPR双激动剂替尔泊肽,GLP-1RA和GIPR拮抗剂AMG133),以期获得更好减重效果。然而,GLP-1以外的肠促胰素靶点在分子作用机制上对减重的贡献仍需大量研究,目前尚无法确定多靶点GLP-1RA类药物的减重效果是否优于单靶点GLP-1RA7。除此之外,单靶点GLP-1RA结构相对简单,机制清晰,更易与基础胰岛素或胰淀素等药物制成固定比例复方制剂。
GLP-1受体介导的不良反应限制GLP-1RA活性增加
提高药物治疗效果的另一种常见手段是提升药物活性。然而,GLP-1RA类药物的活性不能无限增加,因为其与GLP-1受体过强的激动可能引起恶心、呕吐等胃肠道不良反应,导致受试者无法耐受。因此,如何在GLP-1RA活性和安全性风险之间找到平衡点,是值得进一步探索的问题。
活性适中的GLP-1RA可以通过提高给药剂量实现更好疗效
尽管低活性可能限制了GLP-1RA激活受体,影响其发挥降糖和减重的效果。但低活性的GLP-1RA在保持耐受性前提下,有望实现更高剂量,从而带来更好疗效,这在GZR18注射液1b/2a期临床研究中得以验证。
临床前研究表明,GZR18注射液的生物活性与GLP-1受体的亲和力较其他GLP-1RA更低。在人血清白蛋白(HSA)存在或不存在的条件下,GZR18注射液与GLP-1受体的亲和力均弱于利拉鲁肽和司美格鲁肽。
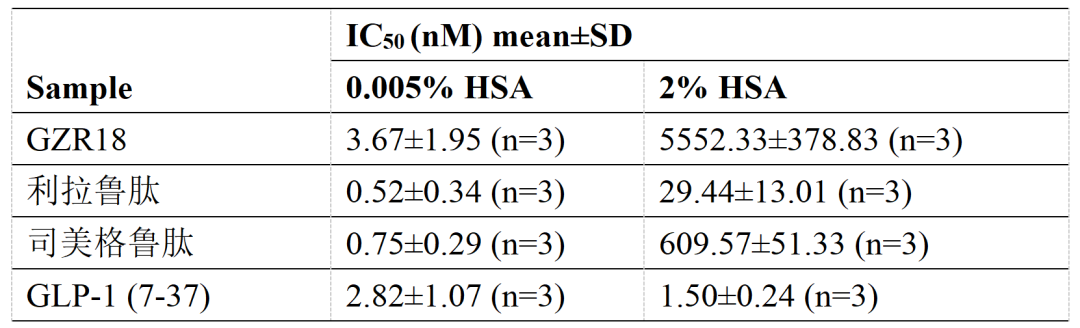
GZR18注射液与GLP-1受体的结合亲和力(IC50)弱于利拉鲁肽、司美格鲁肽和GLP-1 (7-37)(人源GLP-1活性代谢物)
基于GZR18注射液独特的分子活性,其在临床研究中表现出良好的耐受性。给予合理的滴定方案,GZR18注射液在1b/2a期研究中的最高给药剂量达30 mg,实现超过18%(安慰剂调整后)的体重降幅。同时安全性和耐受性良好,不良事件类型和发生率与GLP-1RA类药物相似。这证明,在合理的剂量滴定方案下,受试者能耐受更高的GZR18注射液给药剂量。
GZR18注射液有望实现每两周给药一次,兼顾疗效与安全性
创新的分子设计使GZR18注射液作用时间显著延长,并能实现更高给药剂量。当GZR18注射液在受试者体内达到稳态后,其血药浓度能在两周内维持在有效治疗范围内,从而在两周一次的给药频率下实现显著减重效果:在中国肥胖受试者中,连续35周每两周一次给药后,受试者体重较基线下降12.8%,且安全性与耐受性良好。
为了进一步验证GZR18注射液每两周给药一次的疗效与安全性,甘李药业正在进行一项多中心、安慰剂对照、随机、双盲的2期临床研究。该研究入组338名超重或肥胖成年受试者,探索更宽的剂量范围和两周给药一次的疗效与安全性。目前,所有受试者均已完成试验,初步数据验证了1b/2a期研究结果,特别是双周给药取得了积极效果。目前,已上市的长效GLP-1RA类药物均为单周制剂,GZR18注射液有望成为全球首款双周制剂,受试者仅需每两周注射一次,有效改善GLP-1RA治疗依从性,开启GLP-1治疗新时代。